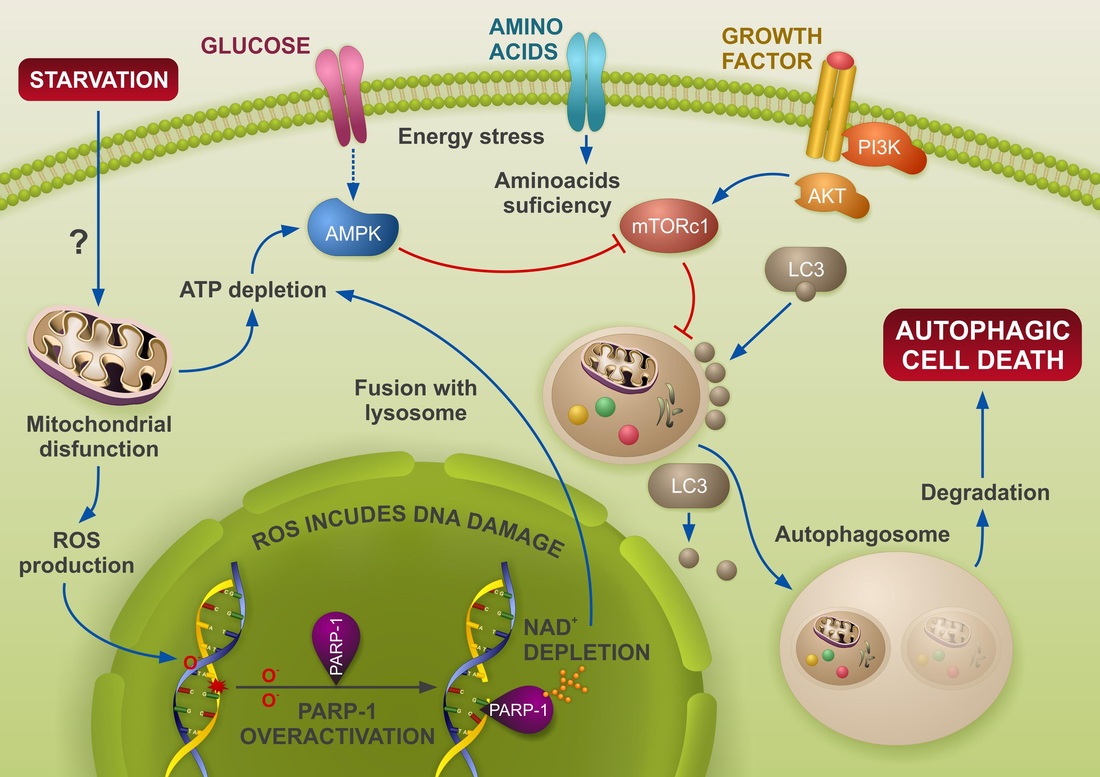
ROS-induced DNA damage and PARP-1 are required for optimal induction of starvation-induced autophagy
In response to nutrient stress, cells start an autophagy program that can lead to adaptation or death. The mechanisms signalling form starvation to the initiation of autophagy are not fully understood. In the current study we show that the absence or inactivation of PARP-1 strongly delays starvation-induced autophagy. We have found that DNA damage is an early event of starvation-induced autophagy as measured by y-H2AX accumulation and comet assay, with PARP-1 knockout cells displaying a reduction in both parameters. During starvation, ROS-induced DNA damage activates PARP-1, leading to ATP depletion (an early event after nutrient deprivation). The absence of PARP-1 blunted AMPK activation and prevented from the complete loss of mTOR activity, leading to a delay in autophagy. PARP depletion favours apoptosis in starved cells, suggesting a pro-survival role of autophagy and PARP-1 activation after nutrient deprivation. In vivo results show that neonates of PARP-1 mutant mice subjected to acute starvation, also display deficient liver autophagy, implying a physiological role for PARP-1 in starvation-induced autophagy. Globally, we have found that PARP signalling pathway is a key regulator of the initial steps of autophagy commitment following starvation.